Same Drug, Different Story
When a pharmaceutical company submits a drug for approval, it doesn't simply resubmit the same documents elsewhere. The same clinical data needs presenting differently for the FDA, the EMA, and Japan's PMDA. Nearly 30% of New Drug Applications aren't approved first time — not because the science is wrong, but because the story told about that science doesn't match what each regulator is looking for.
Something I didn't fully appreciate until recently: when a pharmaceutical company submits a drug for approval in one country, it doesn't simply resubmit the same documents elsewhere. Each health authority has its own requirements, its own formatting expectations, its own evolving rules. The same clinical data might need presenting differently for the FDA, the EMA, and Japan's PMDA — and every adaptation introduces the possibility that something, somewhere, no longer quite matches.
For an industry where a single inconsistency can delay an approval by months, that's a remarkable amount of weight resting on human vigilance.
The eCTD Promise
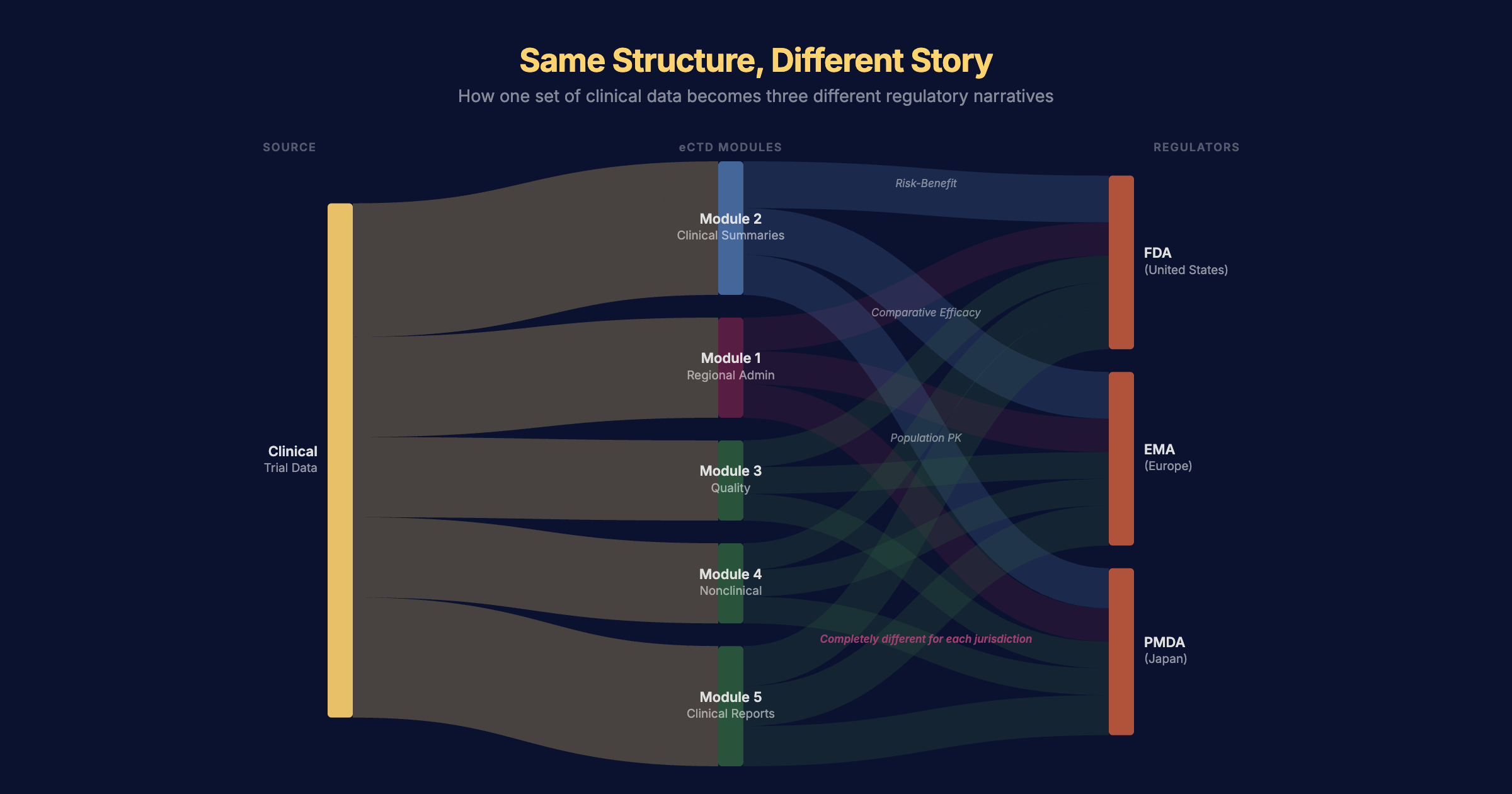
The electronic Common Technical Document — the eCTD — was designed to solve this. Introduced over twenty years ago, it standardised the structure of regulatory submissions across major markets. Over 8 million eCTD submissions have passed through the FDA alone since 2003, and by 2022, 94% of all filings used the format (eCTD Pharma, 2025). It was a genuine step forward.
But standardised structure isn't the same as standardised content. Module 1 — the regional administrative section — is different for every jurisdiction. The clinical narratives in Module 2 might need to emphasise risk-benefit analysis for the FDA, comparative efficacy for the EMA, or local population pharmacokinetics for Japan's PMDA. The underlying science doesn't change, but the story told about that science does. Every time.
Where Things Get Expensive
This is where things get expensive. A study of 825 New Drug Applications found that nearly 30% weren't approved in the first review cycle (Lu et al., 2019). Avalere Health reported that 37% of applications submitted between 2018 and 2022 received a Complete Response Letter — the FDA's way of saying "not yet." Among the common reasons: inconsistencies in data presentation and failure to address previous regulatory questions comprehensively (PharmaSource, 2025).
These aren't scientific failures. They're storytelling failures — the wrong emphasis, the missed cross-reference, the number in one section that doesn't quite match the number in another. In a 100,000-page dossier, that's not carelessness. It's the inevitable consequence of asking human beings to maintain perfect consistency across documents that are being simultaneously adapted for multiple audiences.
A Contextual Problem
Working with regulated organisations across different sectors, I keep coming back to the same observation: the most expensive problems tend to be contextual, not technical. It's rarely that the data doesn't exist. It's that the data isn't presented with enough awareness of who's reading it and what they're looking for.
That's a problem that interests us at Context is Everything, because it's fundamentally about knowledge management — specifically, about ensuring that institutional understanding of what regulators need, what's worked before, and what's changed since the last submission doesn't evaporate between projects or when key people move on.
The pharma industry calls this "regulatory intelligence." We'd call it contextual awareness. Different language, same underlying challenge. And it's one where AI that understands context — rather than simply processes documents — has genuine potential to help.
Whether that help comes from within the industry or from patterns recognised across sectors is an open question.
Probably both.
The Ask
We're looking for partners in the pharmaceutical sector to explore whether our approach to context-aware AI translates to this industry — in the way we believe it does.
Our model is straightforward: we start with a proof of value using your actual data and challenges. If it works, we talk about what comes next. If it doesn't, we'll say so.
The firms that do this successfully treat regulatory knowledge as a private competitive moat — and the expert judgement that makes it work is exactly what AI cannot replace, only amplify.
If that sounds like a conversation worth having, we'd like to hear from you.
Related Articles
Another Industry That Knows Everything But Can't Find Anything
Pharmaceutical regulatory affairs is a $10 billion industry that can map a human genome in hours but still assembles regulatory submissions largely by hand. Nearly 30% of New Drug Applications aren't approved first time — not because of bad science, but because institutional knowledge walks out the door when people move on.
Why Transformations Fail the Departure Test
If everyone who built this left tomorrow, would it keep working? Most transformations fail the departure test because they're built on people, not infrastructure. Here's the knowledge architecture fix.
Why Most AI Projects Fail (And What the 5% Do Differently)
MIT's Project NANDA found 95% of enterprise AI pilots deliver zero return. Companies have invested £30-40 billion with nothing to show. But 5% achieve rapid revenue acceleration. The difference isn't the technology - it's implementation and context.