Another Industry That Knows Everything But Can't Find Anything
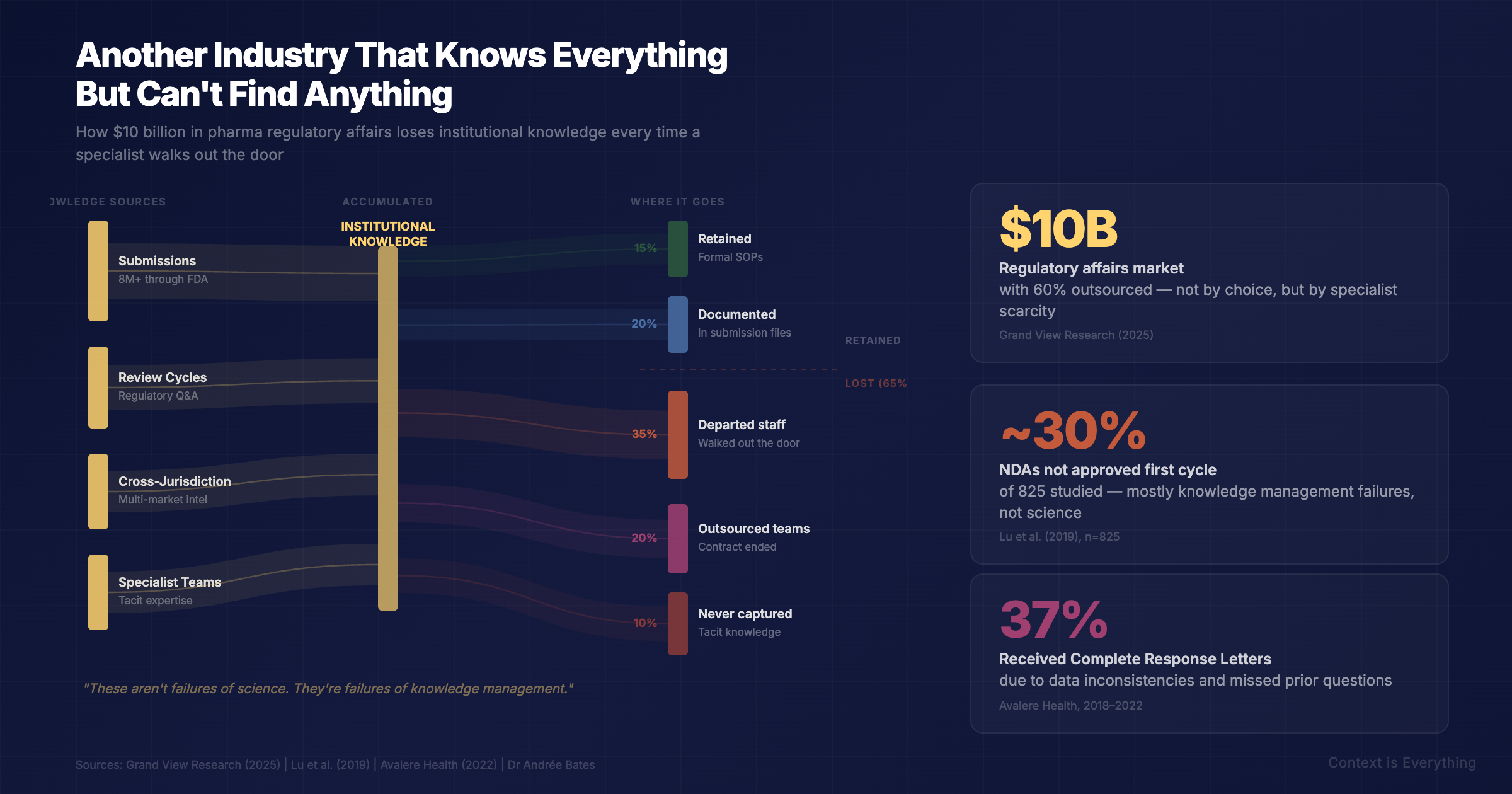
Pharmaceutical regulatory affairs is a $10 billion industry that can map a human genome in hours but still assembles regulatory submissions largely by hand. Nearly 30% of New Drug Applications aren't approved first time — not because of bad science, but because institutional knowledge walks out the door when people move on.
A colleague recently shared Dr Andrée Bates' article on AI in pharmaceutical regulatory affairs, and one line stuck with me: she described the regulatory function as having shifted from strategic partner to "a costly, labour-intensive manual exercise of reconciliation." What struck me wasn't the AI discussion — it was the picture she painted of an industry that can map a human genome in hours but still assembles its regulatory submissions largely by hand.
Working across regulated sectors, I've seen this pattern before. The knowledge is there. The infrastructure to manage it just hasn't caught up.
Pharmaceutical regulatory affairs is a $10 billion industry (Grand View Research, 2025), with around 60% of the work outsourced — not because companies want to outsource, but because they can't recruit or retain enough specialists to manage the volume internally. Over 8 million electronic submissions have passed through the FDA alone since 2003. Each one represents thousands of pages of clinical data, manufacturing records, and safety evidence, assembled and cross-referenced by teams of specialists working across time zones and jurisdictions.
The interesting thing isn't the scale. It's what happens to the knowledge afterwards.
Once a submission is filed, the institutional understanding that went into it — why certain data was presented a particular way, which regulatory questions were anticipated, what worked in a previous jurisdiction — largely stays in people's heads. When those people move on, the knowledge goes with them. The next team starts from scratch, or close to it.
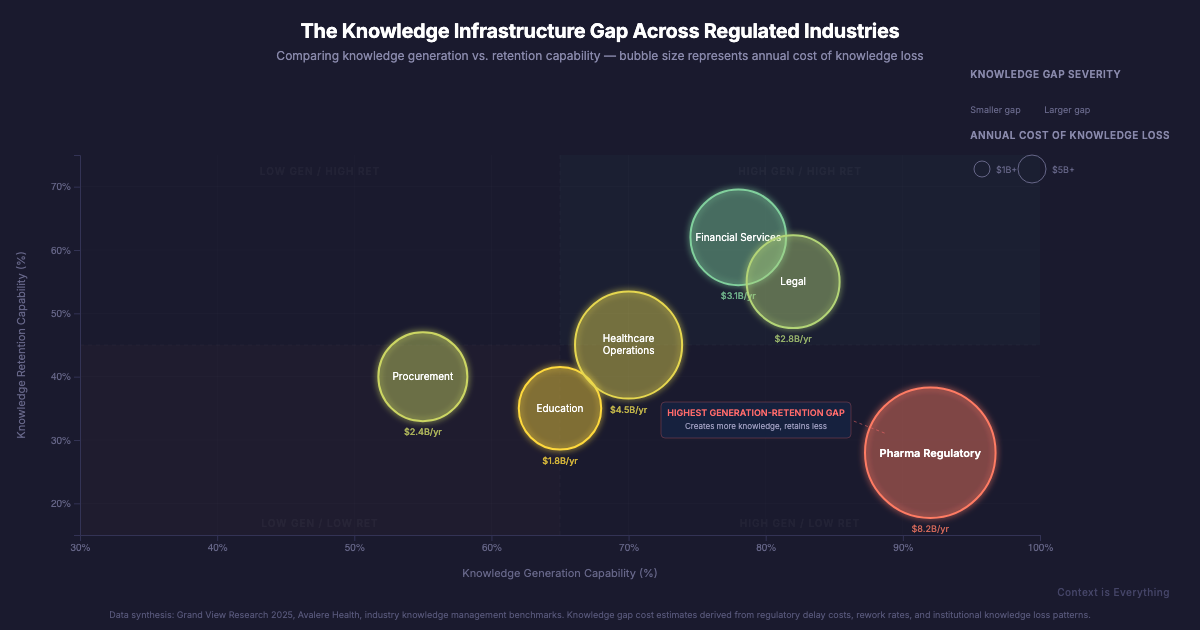
This isn't unique to pharma. We see it in financial services, in education, in procurement. Organisations that are brilliant at generating insight but surprisingly poor at making that insight accessible to the people who need it next. The sector-specific terminology changes. The underlying problem doesn't.
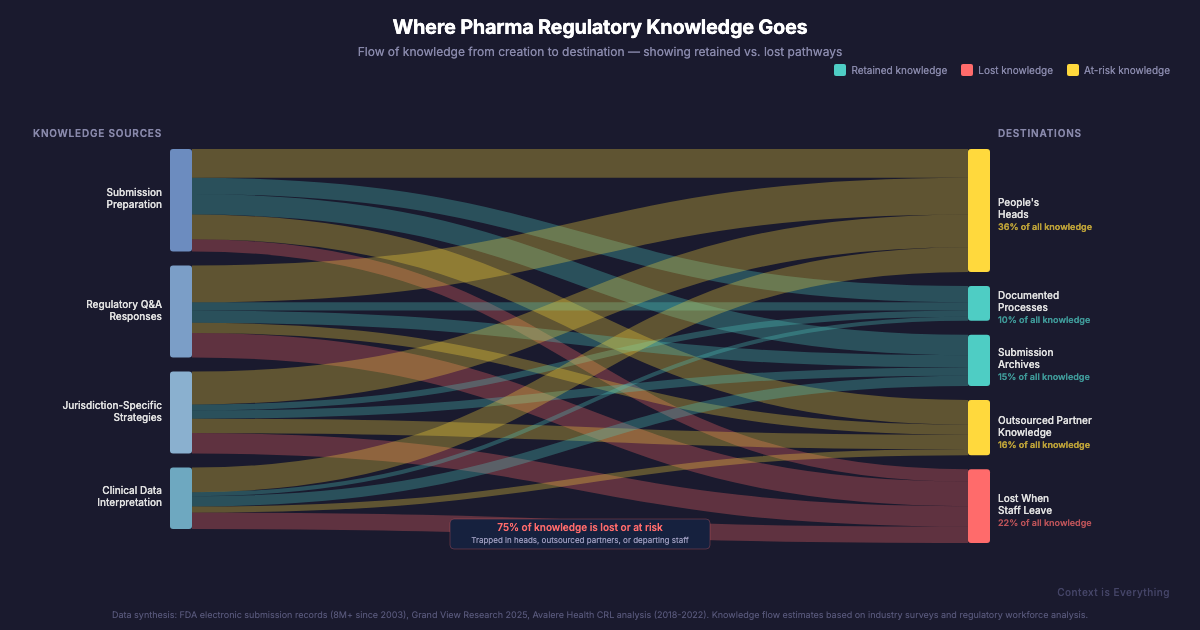
What makes pharma particularly interesting is the cost of getting it wrong. According to a study of 825 New Drug Applications (Lu et al., 2019), nearly 30% weren't approved in the first review cycle. Avalere Health found that 37% of applications submitted between 2018 and 2022 received a Complete Response Letter — essentially a detailed explanation of why the submission couldn't be approved as submitted. Common reasons include inconsistencies in data presentation and failure to address previous regulatory questions comprehensively.
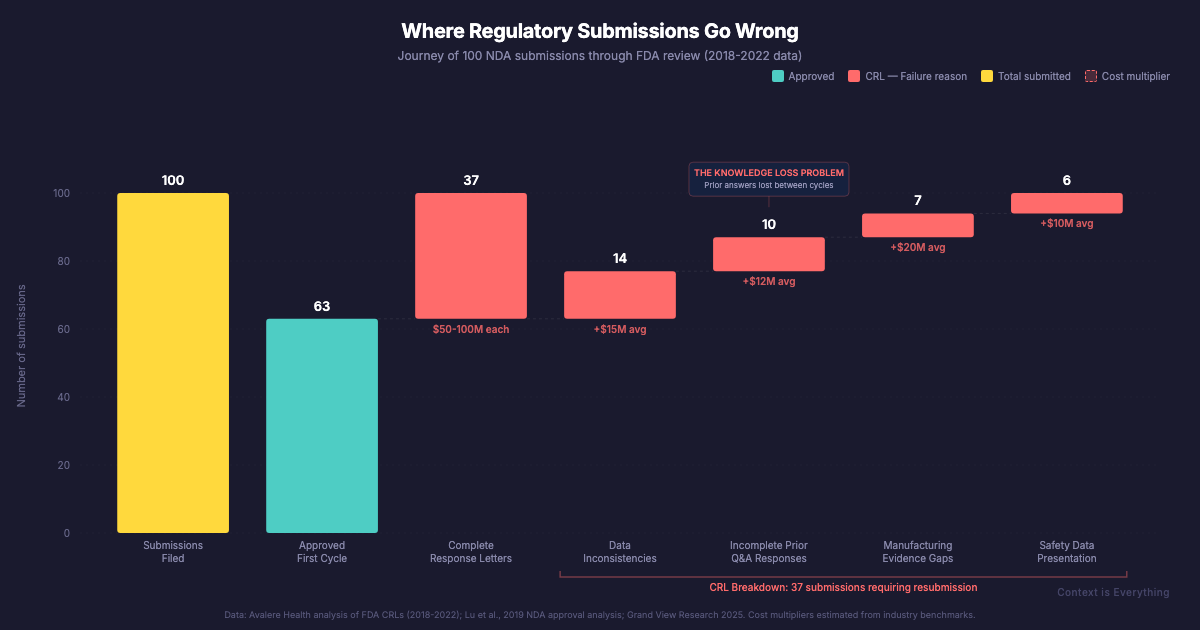
These aren't failures of science. They're failures of knowledge management.
The question worth asking isn't whether AI can help — that much seems clear. It's whether the industry will treat this as a technology problem or recognise it for what it actually is: a knowledge infrastructure problem that technology happens to be well-suited to solving.
There's a meaningful difference between the two. One leads to buying more tools. The other leads to rethinking how institutional knowledge flows through an organisation.
The firms building durable positions in this space are doing so through private data and custom tooling that compounds as base models improve. Keeping that knowledge inside the firm is exactly what private AI deployment enables.
We're looking for partners in the pharmaceutical sector to explore whether our approach to context-aware AI translates to this industry — in the way we believe it does.
Our model is straightforward: we start with a proof of value using your actual data and challenges. If it works, we talk about what comes next. If it doesn't, we'll say so.
If that sounds like a conversation worth having, we'd love to hear from you.
Related Articles
Why Transformations Fail the Departure Test
If everyone who built this left tomorrow, would it keep working? Most transformations fail the departure test because they're built on people, not infrastructure. Here's the knowledge architecture fix.
Identifying High-ROI Processes for AI Automation
Most people intuitively know which tasks are too complex, too arduous, or too boring for humans alone. We've found high-value processes fall into three categories — and picking one from each is the fastest way to prove AI value.
Why Most AI Projects Fail (And What the 5% Do Differently)
MIT's Project NANDA found 95% of enterprise AI pilots deliver zero return. Companies have invested £30-40 billion with nothing to show. But 5% achieve rapid revenue acceleration. The difference isn't the technology - it's implementation and context.